The pathophysiology of acquired/immune-mediated thrombotic thrombocytopenic purpura (aTTP/iTTP) poses a triple threat2-7:
Autoantibody formation

Autoantibody formation leading to ADAMTS13 inhibition resulting in uncleaved vWF (ULvWF)
Microthrombi-driven risks caused by ULvWF binding to platelets to form ULvWF-platelet aggregates
PEX and immunosuppressive therapy directly address only 2 of the 3 aspects of the disease.2,4,5,6,8
The triple threat of aTTP/iTTP deserves a triple therapy regimen with CABLIVI, PEX, and immunosuppressive therapy1-8
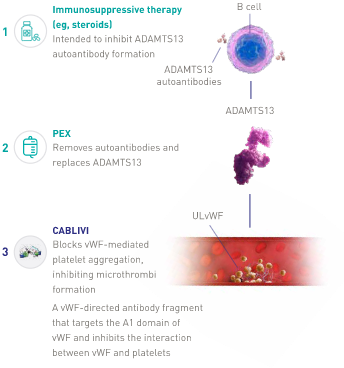
CABLIVI MOA: The first of its kind
Acquired thrombotic thrombocytopenic purpura, or aTTP, is a rare, rapidly progressing, life-threatening autoimmune disease. Left untreated, up to 90% of aTTP patients die.
Historical treatment includes plasma exchange and immunosuppressive therapy, which has improved prognosis. However, despite these treatments, the mortality rate remains high at 8 to 20%. In a large retrospective study, the median time from diagnosis to death was 9 days despite treatment with plasma exchange.
Symptoms of aTTP can be complex and variable. Most common signs include thrombocytopenia, microangiopathic hemolytic anemia, and organ ischemia, resulting from microvascular occlusion. Clinical manifestations of organ ischemia can include strokes, transient ischemic attacks, myocardial infarction, and renal and mesenteric ischemia.
aTTP is caused by the severe deficiency of a von Willebrand factor-cleaving protease, ADAMTS13. This deficiency is due to formation of autoantibodies directed against ADAMTS13. Normally, ADAMTS13 breaks apart ultra-large strings of von Willebrand factor, known as vWF, into smaller pieces. In aTTP patients, however, ADAMTS13 is blocked by autoantibodies, thereby greatly increasing the amount of ultra-large vWF in circulation. Thousands of circulating platelets bind to these ultra-large vWF strings, forming platelet-rich blood clots within small blood vessels throughout the body. These small clots are known as microthrombi.
As of February 2019, CABLIVI became the newest treatment approved by the FDA for aTTP in combination with plasma exchange and immunosuppressive therapy. Plasma exchange removes vWF and autoantibodies and replenishes ADAMTS13. Immunosuppressive therapies are intended to inhibit anti-ADAMTS13 autoantibody production. Neither directly affects the binding of platelets to vWF.
CABLIVI is a monoclonal antibody fragment that was developed to inhibit microthrombi formation in aTTP by binding specifically to the A1 domain of vWF. CABLIVI blocks the interaction between vWF and platelets, thereby reducing both vWF-mediated platelet adhesion and platelet consumption. This inhibits the formation of microthrombi while the underlying autoimmune disease is treated with immunosuppressive therapy.
Choose CABLIVI on day 1 in combination with PEX and immunosuppressive therapy
ISTH Guideline recommendations*
Who should not start CABLIVI?
- CABLIVI is contraindicated in patients with a previous severe hypersensitivity reaction to caplacizumab-yhdp or to any of its excipients
- Withhold CABLIVI treatment 7 days prior to elective surgery, dental procedures, or other invasive interventions
*A conditional recommendation defined as desirable effects of the recommendation probably outweighing the undesirable effects. Assumes timely access to ADAMTS13 testing and clinical diagnosis based on high likelihood of aTTP/iTTP. In de novo patients where no reasonable access to ADAMTS13 activity testing is available, the Guidelines do not recommend CABLIVI; however, treatment of a patient previously diagnosed with aTTP/iTTP could be safely undertaken on clinical grounds without the need for a confirmatory ADAMTS13 test.9
ADAMTS13=a disintegrin and metalloproteinase with a thrombospondin type 1 motif, 13; ISTH=International Society on Thrombosis and Haemostasis; MOA=mechanism of action; PEX=plasma exchange; TTP=thrombotic thrombocytopenic purpura; ULvWF=ultra-large von Willebrand factor; vWF=von Willebrand factor.
IMPORTANT SAFETY INFORMATION AND INDICATIONS
IMPORTANT SAFETY INFORMATION AND INDICATIONS
IMPORTANT SAFETY INFORMATION AND INDICATIONS
CONTRAINDICATIONS:
CABLIVI is contraindicated in patients with a previous severe hypersensitivity reaction to caplacizumab-yhdp or to any of its excipients. Hypersensitivity reactions have included urticaria.
WARNINGS AND PRECAUTIONS:
Hemorrhage:
- CABLIVI increases the risk of bleeding. In clinical studies, severe bleeding adverse reactions of epistaxis, gingival bleeding, upper gastrointestinal hemorrhage, and metrorrhagia were each reported in 1% of subjects. Overall, bleeding events occurred in approximately 58% of patients on CABLIVI versus 43% of patients on placebo.
- In the postmarketing setting cases of life-threatening and fatal bleeding were reported in patients receiving CABLIVI.
- The risk of bleeding is increased in patients with underlying coagulopathies (e.g. hemophilia, other coagulation factor deficiencies). It is also increased with concomitant use of CABLIVI with drugs affecting hemostasis and coagulation.
- Avoid concomitant use of CABLIVI with antiplatelet agents or anticoagulants. If clinically significant bleeding occurs, interrupt use of CABLIVI. Von Willebrand factor concentrate may be administered to rapidly correct hemostasis. If CABLIVI is restarted, monitor closely for signs of bleeding.
- Withhold CABLIVI for 7 days prior to elective surgery, dental procedures or other invasive interventions. If emergency surgery is needed, the use of von Willebrand factor concentrate may be considered to correct hemostasis. After the risk of surgical bleeding has resolved, and CABLIVI is resumed, monitor closely for signs of bleeding.
ADVERSE REACTIONS:
The most common adverse reactions (>15% of patients) were epistaxis (29%), headache (21%) and gingival bleeding (16%).
CONCOMITANT USE OF ANTICOAGULANTS OR ANTIPLATELET AGENTS:
Concomitant use of CABLIVI with any anticoagulant or antiplatelet agent may increase the risk of bleeding. Avoid concomitant use when possible. Assess and monitor closely for bleeding with concomitant use.
PREGNANCY:
There are no available data on CABLIVI use in pregnant women to inform a drug associated risk of major birth defects and miscarriage.
-
Fetal/neonatal adverse reactions: CABLIVI may increase the risk of bleeding in the fetus and neonate. Monitor neonates for bleeding.
-
Maternal adverse reactions: All patients receiving CABLIVI, including pregnant women, are at risk for bleeding. Pregnant women receiving CABLIVI should be carefully monitored for evidence of excessive bleeding.
INDICATIONS:
CABLIVI (caplacizumab-yhdp) is indicated for the treatment of adult patients with acquired thrombotic thrombocytopenic purpura (aTTP), in combination with plasma exchange and immunosuppressive therapy.
Please see full Prescribing Information.
Instructions For Use
Sharps Medical Waste Disposal (PDF)
Learn more about Sanofi’s commitment to fighting counterfeit drugs.
References: 1. CABLIVI. Prescribing information. Genzyme Corporation. 2. Scully M, Cataland SR, Peyvandi F, et al; HERCULES Investigators. Caplacizumab treatment for acquired thrombotic thrombocytopenic purpura. N Engl J Med. 2019;380(4):335-346. doi:10.1056/NEJMoa1806311 3. Joly BS, Coppo P, Veyradier A. Thrombotic thrombocytopenic purpura. Blood. 2017;129(21):2836-2846. doi:10.1182/blood-2016-10-709857 4. Scully M, Hunt BJ, Benjamin S, et al; British Committee for Standards in Haematology. Guidelines on the diagnosis and management of thrombotic thrombocytopenic purpura and other thrombotic microangiopathies. Br J Haematol. 2012;158(3):323-335. doi:10.1111/j.1365-2141.2012.09167.x 5. Kremer Hovinga JA, Coppo P, Lämmle B, Moake JL, Miyata T, Vanhoorelbeke K. Thrombotic thrombocytopenic purpura. Nat Rev Dis Primers. 2017;3:17020. doi:10.1038/nrdp.2017.20 6. Holz J-B. The TITAN trial—assessing the efficacy and safety of an anti-von Willebrand factor Nanobody in patients with acquired thrombotic thrombocytopenic purpura. Transfus Apher Sci. 2012;46(3):343-346. doi:10.1016/j.transci.2012.03.027 7. Azoulay E, Bauer PR, Mariotte E, et al; Nine-i Investigators. Expert statement on the ICU management of patients with thrombotic thrombocytopenic purpura. Intensive Care Med. 2019;45(11):1518-1539. doi:10.1007/s00134-019-05736-5 8. Sayani FA, Abrams CS. How I treat refractory thrombotic thrombocytopenic purpura. Blood. 2015;125(25):3860-3867. doi:10.1182/blood-2014-11-551580 9. Zheng XL, Vesely SK, Cataland SR, et al. ISTH guidelines for the diagnosis of thrombotic thrombocytopenic purpura. J Thromb Haemost. 2020;18(10):2486-2495. doi:10.1111/jth.15006
IMPORTANT SAFETY INFORMATION AND INDICATIONS
CONTRAINDICATIONS:
CABLIVI is contraindicated in patients with a previous severe hypersensitivity reaction to caplacizumab-yhdp or to any of its excipients. Hypersensitivity reactions have included urticaria.
WARNINGS AND PRECAUTIONS:
Hemorrhage:
- CABLIVI increases the risk of bleeding. In clinical studies, severe bleeding adverse reactions of epistaxis, gingival bleeding, upper gastrointestinal hemorrhage, and metrorrhagia were each reported in 1% of subjects. Overall, bleeding events occurred in approximately 58% of patients on CABLIVI versus 43% of patients on placebo.
- In the postmarketing setting cases of life-threatening and fatal bleeding were reported in patients receiving CABLIVI.
- The risk of bleeding is increased in patients with underlying coagulopathies (e.g. hemophilia, other coagulation factor deficiencies). It is also increased with concomitant use of CABLIVI with drugs affecting hemostasis and coagulation.
- Avoid concomitant use of CABLIVI with antiplatelet agents or anticoagulants. If clinically significant bleeding occurs, interrupt use of CABLIVI. Von Willebrand factor concentrate may be administered to rapidly correct hemostasis. If CABLIVI is restarted, monitor closely for signs of bleeding.
- Withhold CABLIVI for 7 days prior to elective surgery, dental procedures or other invasive interventions. If emergency surgery is needed, the use of von Willebrand factor concentrate may be considered to correct hemostasis. After the risk of surgical bleeding has resolved, and CABLIVI is resumed, monitor closely for signs of bleeding.
ADVERSE REACTIONS:
The most common adverse reactions (>15% of patients) were epistaxis (29%), headache (21%) and gingival bleeding (16%).
CONCOMITANT USE OF ANTICOAGULANTS OR ANTIPLATELET AGENTS:
Concomitant use of CABLIVI with any anticoagulant or antiplatelet agent may increase the risk of bleeding. Avoid concomitant use when possible. Assess and monitor closely for bleeding with concomitant use.
PREGNANCY:
There are no available data on CABLIVI use in pregnant women to inform a drug associated risk of major birth defects and miscarriage.
-
Fetal/neonatal adverse reactions: CABLIVI may increase the risk of bleeding in the fetus and neonate. Monitor neonates for bleeding.
-
Maternal adverse reactions: All patients receiving CABLIVI, including pregnant women, are at risk for bleeding. Pregnant women receiving CABLIVI should be carefully monitored for evidence of excessive bleeding.
INDICATIONS:
CABLIVI (caplacizumab-yhdp) is indicated for the treatment of adult patients with acquired thrombotic thrombocytopenic purpura (aTTP), in combination with plasma exchange and immunosuppressive therapy.
Please see full Prescribing Information.
Instructions For Use
Sharps Medical Waste Disposal (PDF)
Learn more about Sanofi’s commitment to fighting counterfeit drugs.
References: 1. CABLIVI. Prescribing information. Genzyme Corporation. 2. Scully M, Cataland SR, Peyvandi F, et al; HERCULES Investigators. Caplacizumab treatment for acquired thrombotic thrombocytopenic purpura. N Engl J Med. 2019;380(4):335-346. doi:10.1056/NEJMoa1806311 3. Joly BS, Coppo P, Veyradier A. Thrombotic thrombocytopenic purpura. Blood. 2017;129(21):2836-2846. doi:10.1182/blood-2016-10-709857 4. Scully M, Hunt BJ, Benjamin S, et al; British Committee for Standards in Haematology. Guidelines on the diagnosis and management of thrombotic thrombocytopenic purpura and other thrombotic microangiopathies. Br J Haematol. 2012;158(3):323-335. doi:10.1111/j.1365-2141.2012.09167.x 5. Kremer Hovinga JA, Coppo P, Lämmle B, Moake JL, Miyata T, Vanhoorelbeke K. Thrombotic thrombocytopenic purpura. Nat Rev Dis Primers. 2017;3:17020. doi:10.1038/nrdp.2017.20 6. Holz J-B. The TITAN trial—assessing the efficacy and safety of an anti-von Willebrand factor Nanobody in patients with acquired thrombotic thrombocytopenic purpura. Transfus Apher Sci. 2012;46(3):343-346. doi:10.1016/j.transci.2012.03.027 7. Azoulay E, Bauer PR, Mariotte E, et al; Nine-i Investigators. Expert statement on the ICU management of patients with thrombotic thrombocytopenic purpura. Intensive Care Med. 2019;45(11):1518-1539. doi:10.1007/s00134-019-05736-5 8. Sayani FA, Abrams CS. How I treat refractory thrombotic thrombocytopenic purpura. Blood. 2015;125(25):3860-3867. doi:10.1182/blood-2014-11-551580 9. Zheng XL, Vesely SK, Cataland SR, et al. ISTH guidelines for the diagnosis of thrombotic thrombocytopenic purpura. J Thromb Haemost. 2020;18(10):2486-2495. doi:10.1111/jth.15006
IMPORTANT SAFETY INFORMATION AND INDICATIONS
IMPORTANT SAFETY INFORMATION AND INDICATIONS
CONTRAINDICATIONS:
CABLIVI is contraindicated in patients with a previous severe hypersensitivity reaction to caplacizumab-yhdp or to any of its excipients. Hypersensitivity reactions have included urticaria.
WARNINGS AND PRECAUTIONS:
Hemorrhage:
- CABLIVI increases the risk of bleeding. In clinical studies, severe bleeding adverse reactions of epistaxis, gingival bleeding, upper gastrointestinal hemorrhage, and metrorrhagia were each reported in 1% of subjects. Overall, bleeding events occurred in approximately 58% of patients on CABLIVI versus 43% of patients on placebo.
- In the postmarketing setting cases of life-threatening and fatal bleeding were reported in patients receiving CABLIVI.
- The risk of bleeding is increased in patients with underlying coagulopathies (e.g. hemophilia, other coagulation factor deficiencies). It is also increased with concomitant use of CABLIVI with drugs affecting hemostasis and coagulation.
- Avoid concomitant use of CABLIVI with antiplatelet agents or anticoagulants. If clinically significant bleeding occurs, interrupt use of CABLIVI. Von Willebrand factor concentrate may be administered to rapidly correct hemostasis. If CABLIVI is restarted, monitor closely for signs of bleeding.
- Withhold CABLIVI for 7 days prior to elective surgery, dental procedures or other invasive interventions. If emergency surgery is needed, the use of von Willebrand factor concentrate may be considered to correct hemostasis. After the risk of surgical bleeding has resolved, and CABLIVI is resumed, monitor closely for signs of bleeding.
ADVERSE REACTIONS:
The most common adverse reactions (>15% of patients) were epistaxis (29%), headache (21%) and gingival bleeding (16%).
CONCOMITANT USE OF ANTICOAGULANTS OR ANTIPLATELET AGENTS:
Concomitant use of CABLIVI with any anticoagulant or antiplatelet agent may increase the risk of bleeding. Avoid concomitant use when possible. Assess and monitor closely for bleeding with concomitant use.
PREGNANCY:
There are no available data on CABLIVI use in pregnant women to inform a drug associated risk of major birth defects and miscarriage.
-
Fetal/neonatal adverse reactions: CABLIVI may increase the risk of bleeding in the fetus and neonate. Monitor neonates for bleeding.
-
Maternal adverse reactions: All patients receiving CABLIVI, including pregnant women, are at risk for bleeding. Pregnant women receiving CABLIVI should be carefully monitored for evidence of excessive bleeding.
INDICATIONS:
CABLIVI (caplacizumab-yhdp) is indicated for the treatment of adult patients with acquired thrombotic thrombocytopenic purpura (aTTP), in combination with plasma exchange and immunosuppressive therapy.
Please see full Prescribing Information.
Instructions For Use
Sharps Medical Waste Disposal (PDF)
Learn more about Sanofi’s commitment to fighting counterfeit drugs.
References: 1. CABLIVI. Prescribing information. Genzyme Corporation. 2. Scully M, Cataland SR, Peyvandi F, et al; HERCULES Investigators. Caplacizumab treatment for acquired thrombotic thrombocytopenic purpura. N Engl J Med. 2019;380(4):335-346. doi:10.1056/NEJMoa1806311 3. Joly BS, Coppo P, Veyradier A. Thrombotic thrombocytopenic purpura. Blood. 2017;129(21):2836-2846. doi:10.1182/blood-2016-10-709857 4. Scully M, Hunt BJ, Benjamin S, et al; British Committee for Standards in Haematology. Guidelines on the diagnosis and management of thrombotic thrombocytopenic purpura and other thrombotic microangiopathies. Br J Haematol. 2012;158(3):323-335. doi:10.1111/j.1365-2141.2012.09167.x 5. Kremer Hovinga JA, Coppo P, Lämmle B, Moake JL, Miyata T, Vanhoorelbeke K. Thrombotic thrombocytopenic purpura. Nat Rev Dis Primers. 2017;3:17020. doi:10.1038/nrdp.2017.20 6. Holz J-B. The TITAN trial—assessing the efficacy and safety of an anti-von Willebrand factor Nanobody in patients with acquired thrombotic thrombocytopenic purpura. Transfus Apher Sci. 2012;46(3):343-346. doi:10.1016/j.transci.2012.03.027 7. Azoulay E, Bauer PR, Mariotte E, et al; Nine-i Investigators. Expert statement on the ICU management of patients with thrombotic thrombocytopenic purpura. Intensive Care Med. 2019;45(11):1518-1539. doi:10.1007/s00134-019-05736-5 8. Sayani FA, Abrams CS. How I treat refractory thrombotic thrombocytopenic purpura. Blood. 2015;125(25):3860-3867. doi:10.1182/blood-2014-11-551580 9. Zheng XL, Vesely SK, Cataland SR, et al. ISTH guidelines for the diagnosis of thrombotic thrombocytopenic purpura. J Thromb Haemost. 2020;18(10):2486-2495. doi:10.1111/jth.15006
IMPORTANT SAFETY INFORMATION AND INDICATIONS
CONTRAINDICATIONS:
CABLIVI is contraindicated in patients with a previous severe hypersensitivity reaction to caplacizumab-yhdp or to any of its excipients. Hypersensitivity reactions have included urticaria.
WARNINGS AND PRECAUTIONS:
Hemorrhage:
- CABLIVI increases the risk of bleeding. In clinical studies, severe bleeding adverse reactions of epistaxis, gingival bleeding, upper gastrointestinal hemorrhage, and metrorrhagia were each reported in 1% of subjects. Overall, bleeding events occurred in approximately 58% of patients on CABLIVI versus 43% of patients on placebo.
- In the postmarketing setting cases of life-threatening and fatal bleeding were reported in patients receiving CABLIVI.
- The risk of bleeding is increased in patients with underlying coagulopathies (e.g. hemophilia, other coagulation factor deficiencies). It is also increased with concomitant use of CABLIVI with drugs affecting hemostasis and coagulation.
- Avoid concomitant use of CABLIVI with antiplatelet agents or anticoagulants. If clinically significant bleeding occurs, interrupt use of CABLIVI. Von Willebrand factor concentrate may be administered to rapidly correct hemostasis. If CABLIVI is restarted, monitor closely for signs of bleeding.
- Withhold CABLIVI for 7 days prior to elective surgery, dental procedures or other invasive interventions. If emergency surgery is needed, the use of von Willebrand factor concentrate may be considered to correct hemostasis. After the risk of surgical bleeding has resolved, and CABLIVI is resumed, monitor closely for signs of bleeding.
ADVERSE REACTIONS:
The most common adverse reactions (>15% of patients) were epistaxis (29%), headache (21%) and gingival bleeding (16%).
CONCOMITANT USE OF ANTICOAGULANTS OR ANTIPLATELET AGENTS:
Concomitant use of CABLIVI with any anticoagulant or antiplatelet agent may increase the risk of bleeding. Avoid concomitant use when possible. Assess and monitor closely for bleeding with concomitant use.
PREGNANCY:
There are no available data on CABLIVI use in pregnant women to inform a drug associated risk of major birth defects and miscarriage.
-
Fetal/neonatal adverse reactions: CABLIVI may increase the risk of bleeding in the fetus and neonate. Monitor neonates for bleeding.
-
Maternal adverse reactions: All patients receiving CABLIVI, including pregnant women, are at risk for bleeding. Pregnant women receiving CABLIVI should be carefully monitored for evidence of excessive bleeding.
INDICATIONS:
CABLIVI (caplacizumab-yhdp) is indicated for the treatment of adult patients with acquired thrombotic thrombocytopenic purpura (aTTP), in combination with plasma exchange and immunosuppressive therapy.
Please see full Prescribing Information.
Instructions For Use
Sharps Medical Waste Disposal (PDF)
Learn more about Sanofi’s commitment to fighting counterfeit drugs.
References: 1. CABLIVI. Prescribing information. Genzyme Corporation. 2. Scully M, Cataland SR, Peyvandi F, et al; HERCULES Investigators. Caplacizumab treatment for acquired thrombotic thrombocytopenic purpura. N Engl J Med. 2019;380(4):335-346. doi:10.1056/NEJMoa1806311 3. Joly BS, Coppo P, Veyradier A. Thrombotic thrombocytopenic purpura. Blood. 2017;129(21):2836-2846. doi:10.1182/blood-2016-10-709857 4. Scully M, Hunt BJ, Benjamin S, et al; British Committee for Standards in Haematology. Guidelines on the diagnosis and management of thrombotic thrombocytopenic purpura and other thrombotic microangiopathies. Br J Haematol. 2012;158(3):323-335. doi:10.1111/j.1365-2141.2012.09167.x 5. Kremer Hovinga JA, Coppo P, Lämmle B, Moake JL, Miyata T, Vanhoorelbeke K. Thrombotic thrombocytopenic purpura. Nat Rev Dis Primers. 2017;3:17020. doi:10.1038/nrdp.2017.20 6. Holz J-B. The TITAN trial—assessing the efficacy and safety of an anti-von Willebrand factor Nanobody in patients with acquired thrombotic thrombocytopenic purpura. Transfus Apher Sci. 2012;46(3):343-346. doi:10.1016/j.transci.2012.03.027 7. Azoulay E, Bauer PR, Mariotte E, et al; Nine-i Investigators. Expert statement on the ICU management of patients with thrombotic thrombocytopenic purpura. Intensive Care Med. 2019;45(11):1518-1539. doi:10.1007/s00134-019-05736-5 8. Sayani FA, Abrams CS. How I treat refractory thrombotic thrombocytopenic purpura. Blood. 2015;125(25):3860-3867. doi:10.1182/blood-2014-11-551580 9. Zheng XL, Vesely SK, Cataland SR, et al. ISTH guidelines for the diagnosis of thrombotic thrombocytopenic purpura. J Thromb Haemost. 2020;18(10):2486-2495. doi:10.1111/jth.15006
IMPORTANT SAFETY INFORMATION AND INDICATIONS
IMPORTANT SAFETY INFORMATION AND INDICATIONS
CONTRAINDICATIONS:
CABLIVI is contraindicated in patients with a previous severe hypersensitivity reaction to caplacizumab-yhdp or to any of its excipients. Hypersensitivity reactions have included urticaria.
WARNINGS AND PRECAUTIONS:
Hemorrhage:
- CABLIVI increases the risk of bleeding. In clinical studies, severe bleeding adverse reactions of epistaxis, gingival bleeding, upper gastrointestinal hemorrhage, and metrorrhagia were each reported in 1% of subjects. Overall, bleeding events occurred in approximately 58% of patients on CABLIVI versus 43% of patients on placebo.
- In the postmarketing setting cases of life-threatening and fatal bleeding were reported in patients receiving CABLIVI.
- The risk of bleeding is increased in patients with underlying coagulopathies (e.g. hemophilia, other coagulation factor deficiencies). It is also increased with concomitant use of CABLIVI with drugs affecting hemostasis and coagulation.
- Avoid concomitant use of CABLIVI with antiplatelet agents or anticoagulants. If clinically significant bleeding occurs, interrupt use of CABLIVI. Von Willebrand factor concentrate may be administered to rapidly correct hemostasis. If CABLIVI is restarted, monitor closely for signs of bleeding.
- Withhold CABLIVI for 7 days prior to elective surgery, dental procedures or other invasive interventions. If emergency surgery is needed, the use of von Willebrand factor concentrate may be considered to correct hemostasis. After the risk of surgical bleeding has resolved, and CABLIVI is resumed, monitor closely for signs of bleeding.
ADVERSE REACTIONS:
The most common adverse reactions (>15% of patients) were epistaxis (29%), headache (21%) and gingival bleeding (16%).
CONCOMITANT USE OF ANTICOAGULANTS OR ANTIPLATELET AGENTS:
Concomitant use of CABLIVI with any anticoagulant or antiplatelet agent may increase the risk of bleeding. Avoid concomitant use when possible. Assess and monitor closely for bleeding with concomitant use.
PREGNANCY:
There are no available data on CABLIVI use in pregnant women to inform a drug associated risk of major birth defects and miscarriage.
-
Fetal/neonatal adverse reactions: CABLIVI may increase the risk of bleeding in the fetus and neonate. Monitor neonates for bleeding.
-
Maternal adverse reactions: All patients receiving CABLIVI, including pregnant women, are at risk for bleeding. Pregnant women receiving CABLIVI should be carefully monitored for evidence of excessive bleeding.
INDICATIONS:
CABLIVI (caplacizumab-yhdp) is indicated for the treatment of adult patients with acquired thrombotic thrombocytopenic purpura (aTTP), in combination with plasma exchange and immunosuppressive therapy.
Please see full Prescribing Information.
Instructions For Use
Sharps Medical Waste Disposal (PDF)
Learn more about Sanofi’s commitment to fighting counterfeit drugs.
References: 1. CABLIVI. Prescribing information. Genzyme Corporation. 2. Scully M, Cataland SR, Peyvandi F, et al; HERCULES Investigators. Caplacizumab treatment for acquired thrombotic thrombocytopenic purpura. N Engl J Med. 2019;380(4):335-346. doi:10.1056/NEJMoa1806311 3. Joly BS, Coppo P, Veyradier A. Thrombotic thrombocytopenic purpura. Blood. 2017;129(21):2836-2846. doi:10.1182/blood-2016-10-709857 4. Scully M, Hunt BJ, Benjamin S, et al; British Committee for Standards in Haematology. Guidelines on the diagnosis and management of thrombotic thrombocytopenic purpura and other thrombotic microangiopathies. Br J Haematol. 2012;158(3):323-335. doi:10.1111/j.1365-2141.2012.09167.x 5. Kremer Hovinga JA, Coppo P, Lämmle B, Moake JL, Miyata T, Vanhoorelbeke K. Thrombotic thrombocytopenic purpura. Nat Rev Dis Primers. 2017;3:17020. doi:10.1038/nrdp.2017.20 6. Holz J-B. The TITAN trial—assessing the efficacy and safety of an anti-von Willebrand factor Nanobody in patients with acquired thrombotic thrombocytopenic purpura. Transfus Apher Sci. 2012;46(3):343-346. doi:10.1016/j.transci.2012.03.027 7. Azoulay E, Bauer PR, Mariotte E, et al; Nine-i Investigators. Expert statement on the ICU management of patients with thrombotic thrombocytopenic purpura. Intensive Care Med. 2019;45(11):1518-1539. doi:10.1007/s00134-019-05736-5 8. Sayani FA, Abrams CS. How I treat refractory thrombotic thrombocytopenic purpura. Blood. 2015;125(25):3860-3867. doi:10.1182/blood-2014-11-551580 9. Zheng XL, Vesely SK, Cataland SR, et al. ISTH guidelines for the diagnosis of thrombotic thrombocytopenic purpura. J Thromb Haemost. 2020;18(10):2486-2495. doi:10.1111/jth.15006
IMPORTANT SAFETY INFORMATION AND INDICATIONS
CONTRAINDICATIONS:
CABLIVI is contraindicated in patients with a previous severe hypersensitivity reaction to caplacizumab-yhdp or to any of its excipients. Hypersensitivity reactions have included urticaria.
WARNINGS AND PRECAUTIONS:
Hemorrhage:
- CABLIVI increases the risk of bleeding. In clinical studies, severe bleeding adverse reactions of epistaxis, gingival bleeding, upper gastrointestinal hemorrhage, and metrorrhagia were each reported in 1% of subjects. Overall, bleeding events occurred in approximately 58% of patients on CABLIVI versus 43% of patients on placebo.
- In the postmarketing setting cases of life-threatening and fatal bleeding were reported in patients receiving CABLIVI.
- The risk of bleeding is increased in patients with underlying coagulopathies (e.g. hemophilia, other coagulation factor deficiencies). It is also increased with concomitant use of CABLIVI with drugs affecting hemostasis and coagulation.
- Avoid concomitant use of CABLIVI with antiplatelet agents or anticoagulants. If clinically significant bleeding occurs, interrupt use of CABLIVI. Von Willebrand factor concentrate may be administered to rapidly correct hemostasis. If CABLIVI is restarted, monitor closely for signs of bleeding.
- Withhold CABLIVI for 7 days prior to elective surgery, dental procedures or other invasive interventions. If emergency surgery is needed, the use of von Willebrand factor concentrate may be considered to correct hemostasis. After the risk of surgical bleeding has resolved, and CABLIVI is resumed, monitor closely for signs of bleeding.
ADVERSE REACTIONS:
The most common adverse reactions (>15% of patients) were epistaxis (29%), headache (21%) and gingival bleeding (16%).
CONCOMITANT USE OF ANTICOAGULANTS OR ANTIPLATELET AGENTS:
Concomitant use of CABLIVI with any anticoagulant or antiplatelet agent may increase the risk of bleeding. Avoid concomitant use when possible. Assess and monitor closely for bleeding with concomitant use.
PREGNANCY:
There are no available data on CABLIVI use in pregnant women to inform a drug associated risk of major birth defects and miscarriage.
-
Fetal/neonatal adverse reactions: CABLIVI may increase the risk of bleeding in the fetus and neonate. Monitor neonates for bleeding.
-
Maternal adverse reactions: All patients receiving CABLIVI, including pregnant women, are at risk for bleeding. Pregnant women receiving CABLIVI should be carefully monitored for evidence of excessive bleeding.
INDICATIONS:
CABLIVI (caplacizumab-yhdp) is indicated for the treatment of adult patients with acquired thrombotic thrombocytopenic purpura (aTTP), in combination with plasma exchange and immunosuppressive therapy.
Please see full Prescribing Information.
Instructions For Use
Sharps Medical Waste Disposal (PDF)
Learn more about Sanofi’s commitment to fighting counterfeit drugs.
References: 1. CABLIVI. Prescribing information. Genzyme Corporation. 2. Scully M, Cataland SR, Peyvandi F, et al; HERCULES Investigators. Caplacizumab treatment for acquired thrombotic thrombocytopenic purpura. N Engl J Med. 2019;380(4):335-346. doi:10.1056/NEJMoa1806311 3. Joly BS, Coppo P, Veyradier A. Thrombotic thrombocytopenic purpura. Blood. 2017;129(21):2836-2846. doi:10.1182/blood-2016-10-709857 4. Scully M, Hunt BJ, Benjamin S, et al; British Committee for Standards in Haematology. Guidelines on the diagnosis and management of thrombotic thrombocytopenic purpura and other thrombotic microangiopathies. Br J Haematol. 2012;158(3):323-335. doi:10.1111/j.1365-2141.2012.09167.x 5. Kremer Hovinga JA, Coppo P, Lämmle B, Moake JL, Miyata T, Vanhoorelbeke K. Thrombotic thrombocytopenic purpura. Nat Rev Dis Primers. 2017;3:17020. doi:10.1038/nrdp.2017.20 6. Holz J-B. The TITAN trial—assessing the efficacy and safety of an anti-von Willebrand factor Nanobody in patients with acquired thrombotic thrombocytopenic purpura. Transfus Apher Sci. 2012;46(3):343-346. doi:10.1016/j.transci.2012.03.027 7. Azoulay E, Bauer PR, Mariotte E, et al; Nine-i Investigators. Expert statement on the ICU management of patients with thrombotic thrombocytopenic purpura. Intensive Care Med. 2019;45(11):1518-1539. doi:10.1007/s00134-019-05736-5 8. Sayani FA, Abrams CS. How I treat refractory thrombotic thrombocytopenic purpura. Blood. 2015;125(25):3860-3867. doi:10.1182/blood-2014-11-551580 9. Zheng XL, Vesely SK, Cataland SR, et al. ISTH guidelines for the diagnosis of thrombotic thrombocytopenic purpura. J Thromb Haemost. 2020;18(10):2486-2495. doi:10.1111/jth.15006
